The pi bond refers to sideways orbital overlap of one of the 3 p orbitals resulting in a shorter bond. It also has 3 individual p orbitals in it each containing 2 electrons.

Solved Draw The Resonance Structure For The Peptide Bond Chegg Com
We are taught that a p orbitals house 6 electrons but this is wrong.
. A double bond 1 pi bond 1 sigma bond. The p sublevel houses 6 electrons.
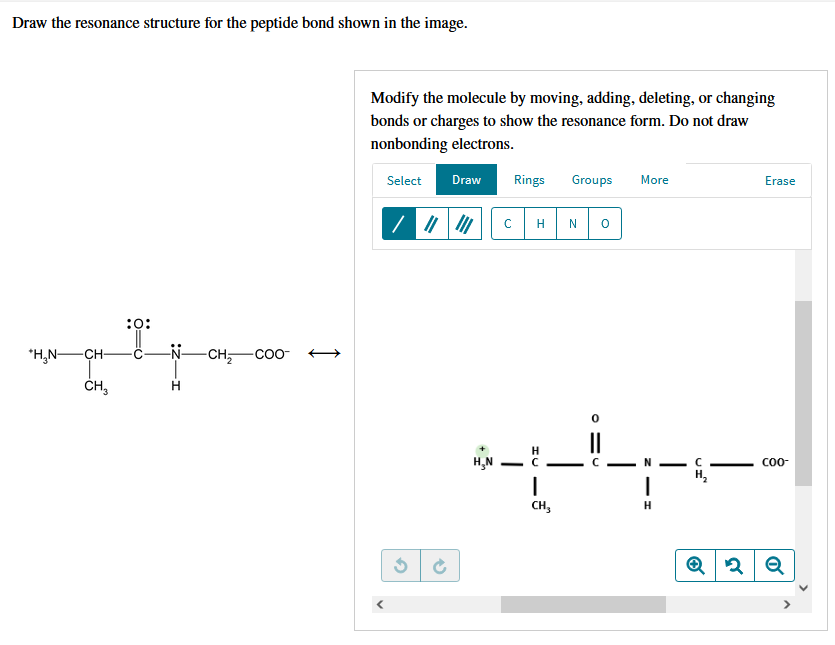
Solved Draw The Resonance Structure For The Peptide Bond Chegg Com
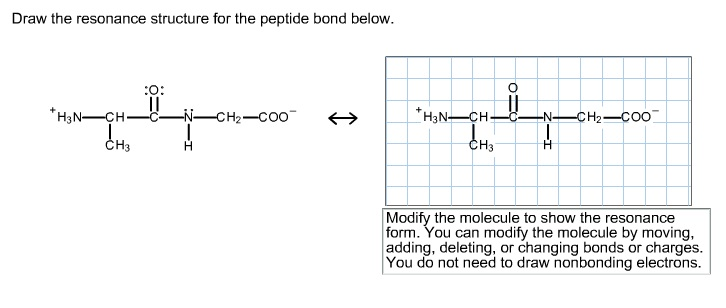
Solved Draw The Resonance Structure For The Peptide Bond Chegg Com
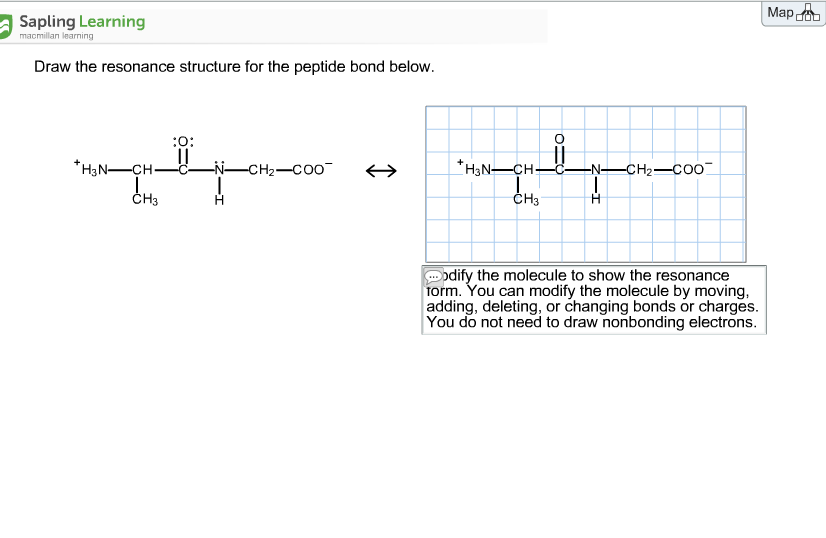
Solved Draw The Resonance Structure For The Peptide Bond Chegg Com
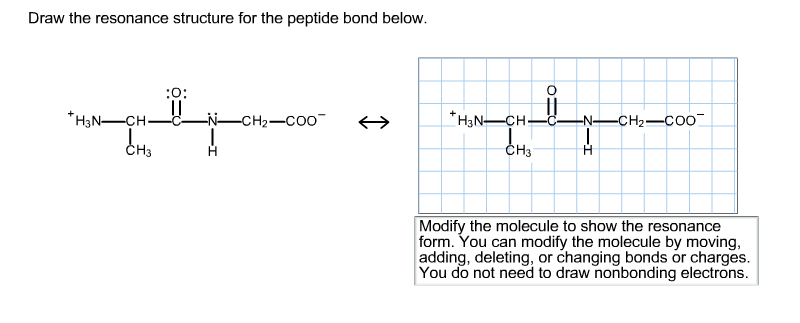
Solved Draw The Resonance Structure For The Peptide Bond Chegg Com

Oneclass Draw The Resonance Structure For The Peptide Bond Below Modify The Molecule To Show The Re
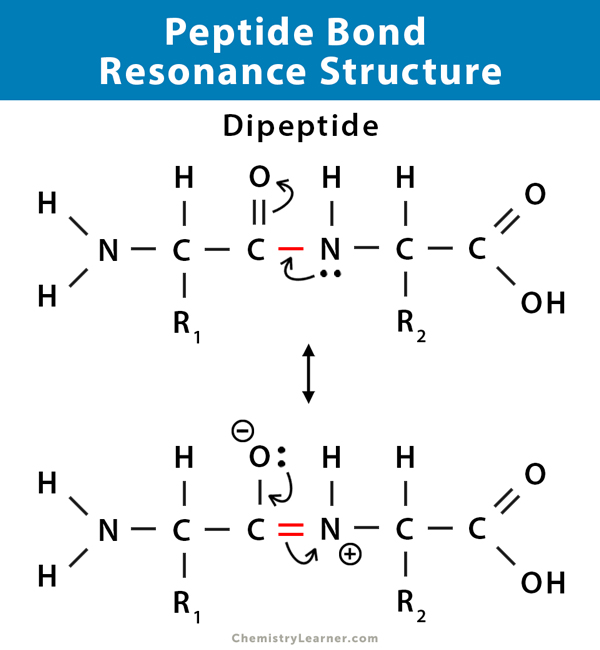
Peptide Bond Definition Structure Mechanism And Examples
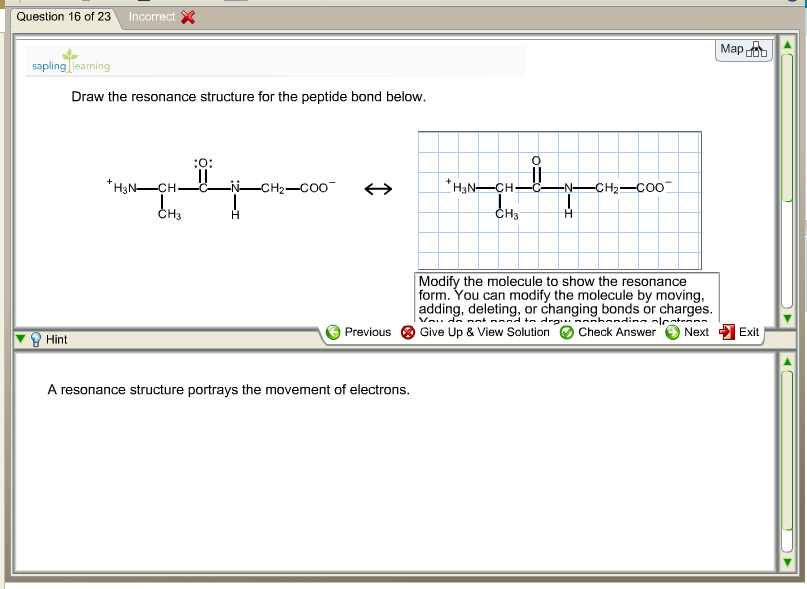
Solved Draw The Resonance Structure For The Peptide Bond Chegg Com
0 comments
Post a Comment